乙肝临床治愈的共识和争议:慢乙肝患者抗病毒治疗停药后能否获得长期受益?
编者按:最近庄辉院士等发表在中华肝脏病杂志的《乙型肝炎临床治愈:共识与争议》[1](相关链接)(以下简称为《共识与争议》)中认可了目前临床治愈是慢乙肝治疗的理想终点,然而在科学工作者不断探索优化抗病毒治疗策略的过程中,专家认为无论是联合核苷(酸)类似物和聚乙二醇干扰素的治疗策略,还是适时终止抗病毒药物治疗,抑或是加速创新药物的研发,目前都充满着机遇与争议。
针对《共识与指南》中提出的临床治愈争议问题,根据慢乙肝临床治愈发展历程及相关研究,肝霖君将推出3篇系列文章,尝试对其中的争议问题进行讨论和分析,第一篇为基于聚乙二醇干扰素α治疗优势人群的相关争议探讨(相关链接)。今天第二篇将就基于慢乙肝患者抗病毒治疗停药后能否获得长期受益进行讨论。

《共识与争议》提出:尽管也有证据表明,适时停药可提高患者HBsAg清除率,但停药是否能给患者带来长期获益仍存在着争议。
慢乙肝患者抗病毒治疗停药的相关研究还是比较多的,我们将分为基于聚乙二醇干扰素α(PEGIFNα)治疗停药和核苷类药物(NA)治疗停药后患者的长期受益来讨论。
一、抗病毒治疗可显著改善慢乙肝患者的远期结局
慢性乙型肝炎防治指南(2019年版)[2]指出:非肝硬化HBV感染者的肝癌年发生率为0.5%-1.0%。而肝硬化患者肝癌年发生率达3%-6%。
以未治疗的慢乙肝患者作为对照组的多项研究显示:未治疗的慢乙肝患者5年肝癌累积发生率高达15%左右[3-5],有肝硬化的甚至高达19.6%[6]。
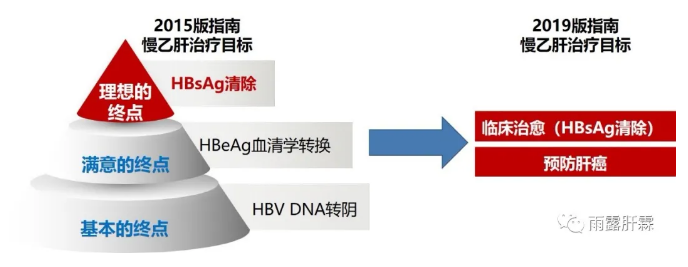
2015年《慢性乙型肝炎防治指南》[7]将慢乙肝的治疗终点分为基本的终点(HBVDNA转阴)、满意的终点(HBeAg血清学转换)及理想的终点(HBsAg清除)。随着抗病毒治疗目标的递进,慢乙肝患者的5年肝癌累积发生率递减,从获得基本终点时的3.6%-11.4%,下降到获得满意终点时的<2.58%,再到获得理想终点时的仅1%左右。
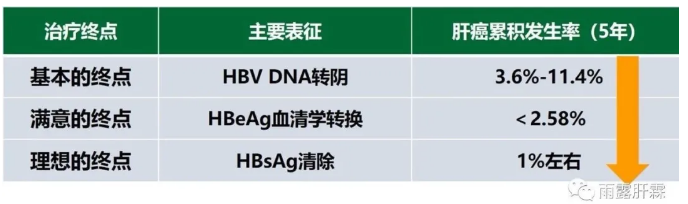
近年来多项研究证实慢乙肝患者获得HBsAg清除后可使肝癌发生风险降到最低。2019年版《慢性乙型肝炎防治指南》中治疗目标中仅强调了追求临床治愈(HBsAg清除)和降低肝癌发生风险,不再将治疗目标分为三个层次。
二、基于PEGIFNα治疗的慢乙肝患者停药后可获得长期获益
01 基于(PEG)IFNα治疗慢乙肝患者停药后持久应答佳
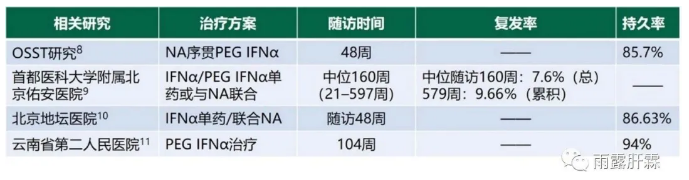
临床治愈是慢乙肝患者抗病毒治疗的理想目标,大量研究证实基于PEGIFNα治疗慢乙肝患者可获得较高的临床治愈率,其中优势患者可达30%-80%。且通过基于(PEG)IFNα治疗获得HBsAg清除的持久性佳,复发风险低[8-11]。
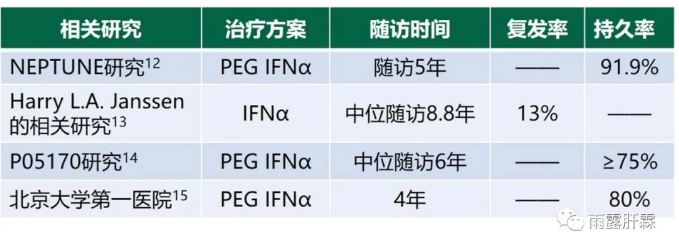
而基于(PEG)IFNα治疗未获得临床治愈的患者,实际在获得治疗应答(HBeAg血清学转换或联合应答)后复发率也低,其长期随访的持久应答率也维持在75%以上[12-15]。
02 基于(PEG)IFNα治疗停药后慢乙肝患者延迟应答情况佳
PEGIFNα治疗慢乙肝的优势在于其免疫调节作用,可使患者获得停药后持久和延迟的免疫学应答。
南方医科大学南方医院的相关研究发现PEGIFNα治疗的慢乙肝患者,治疗结束时未获得HBeAg血清学应答和联合应答的患者中分别有18.2%和15.4%在随访期间获得了HBeAg血清学应答率和联合应答率[14]。
OSST研究中慢乙肝患者基于PEGIFNα治疗48周后随访1年,发现HBeAg血清学转换率从治疗结束时的17.7%提高至38.7%[8]。
韩国最新研究纳入122例完成完整PEGIFNα治疗疗程的患者,其中7.4%(8/122)的患者获得了停药后HBsAg清除[16]。
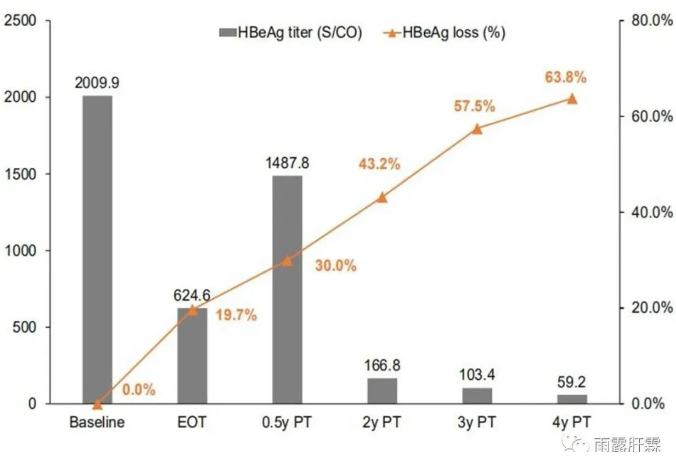
北京大学第一医院牵头的相关研究4年长期随访结果显示PEGIFNα治疗初治HBeAg阳性慢乙肝患者HBeAg水平从治疗结束时的624.6S/CO显著下降到随访4年后的59.2S/CO,HBeAg清除率从治疗结束时的19.7%显著增加到随访4年后的63.8%。33.9%的无应答者在随访4年期间出现延迟应答[15]。
03 基于PEGIFNα治疗的慢乙肝患者远期不良结局风险极低
基于PEGIFNα的优化治疗方式可大大提高慢乙肝患者HBsAg清除率,且显著高于NA。而HBsAg清除可将慢乙肝患者远期不良结局发生风险最小化[16-23],肝霖君曾系统综述HBsAg清除患者的肝癌发生风险极低(相关链接)。
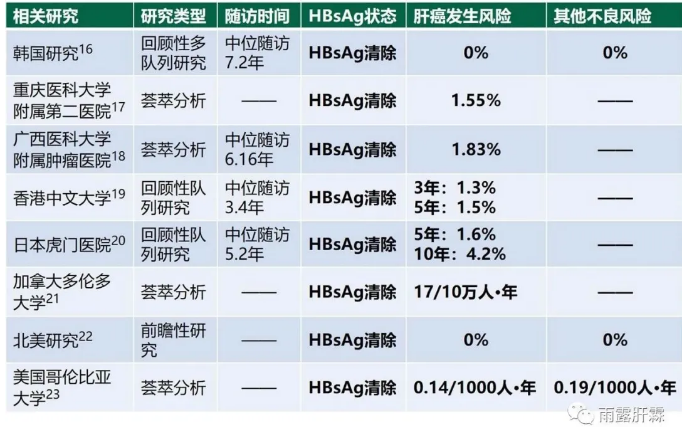
另外,也有研究证实:采用PEGIFNα治疗的患者,即便是未获得HBsAg清除,其肝组织学均有显著改善[16,24],且肝癌发生风险基本与获得HBsAg清除的患者相似(如重庆医科大学附属第二医院[25]、上海瑞金医院[26]和台湾研究[27]等分析PEGIFNα整体治疗后肝癌风险,均未区分患者是否获得HBsAg清除)。
三、基于NA治疗慢乙肝患者停药后长期受益与否仍需权衡
01 NA治疗的慢乙肝患者停药后复发风险高
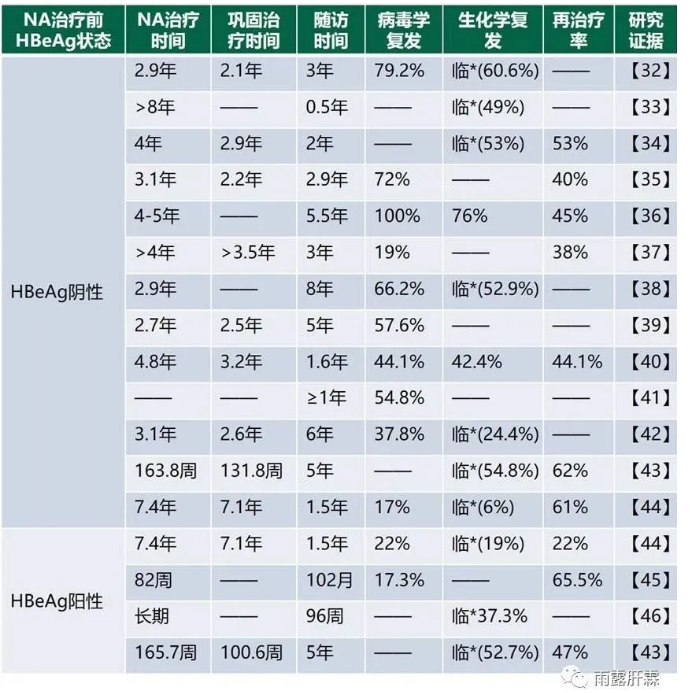
无论是基于PEGIFNα还是NA治疗获得HBsAg清除后的持久性也同样很好[28-31],但NA治疗HBsAg清除率极低,且停药后复发率高。中国《慢性乙型肝炎防治指南(2019年版)》指出对于HBeAg阳性慢乙肝患者NA治疗1年若HBVDNA低于检测下限、ALT复常和HBeAg血清学转换后,再巩固治疗至少3年(每隔6个月复查1次)仍保持不变,可考虑停药,延长疗程可减少复发,而对于HBeAg阴性慢乙肝患者NA治疗后建议HBsAg消失且HBVDNA检测不到后停药随访。因此很多专家并不建议NA停药,认为应该长期服用。
然而无论是HBeAg阴性[32-44]还是HBeAg阳性[43-46]慢乙肝患者NA治疗停药后,若未获得HBsAg清除,复发风险极高(相关链接一、相关链接二)。
02 慢乙肝患者NA停药后能否追求HBsAg清除?
HBeAg阴性慢乙肝患者NA停药后虽然面临着复发的高风险,但是仍有研究发现,这部分人群仍然有着较高的HBsAg清除机会。如何判断这部分人群会复发还是将会获得更高的临床治愈机会,肝霖君也曾系统综述相关内容(相关链接)。
“宿主主导型”和“病毒主导型”肝炎类型对于预测NA停药后结局有重要意义,因此区分“宿主主导型”和“病毒主导型”肝炎类型,是拟定再治疗决策的关键。联合HBsAg和ALT水平更好地区分病毒主导型肝炎和宿主主导型肝炎:HBsAg较前一水平(在ALT升至峰值前或接近峰值时)下降>10%反映了宿主正在进行有效的HBV免疫清除(“宿主主导型肝炎”);反之,如果HBsAg随着ALT的升高而持续增加,或者在ALT峰值后仍保持高水平,则反映了宿主免疫清除的失败(“病毒主导型肝炎”)。对于“病毒主导型肝炎”患者而言,需要再治疗,若可联合PEGIFNα治疗,仍然有机会获得HBsAg清除。
而“宿主主导型肝炎”患者,可暂不治疗,但需要密切监测生化指标,避免病情加重,以追求远期受益。
四、抗病毒治疗均可降低慢乙肝患者肝癌发生风险,PEGIFNα优于NA
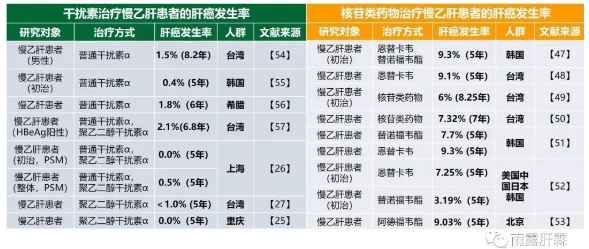
然而根据已发表的研究,慢乙肝患者经NA治疗,肝癌累积发生率相比不治疗有显著降低,但依然处于较高水平[47-53]。
2020年发表的两项研究表明:慢乙肝患者经恩替卡韦或替诺福韦酯长期治疗后的肝癌发生风险仍稳定存在,治疗前5年和5年后的肝癌发生率无显著变化,5年的肝癌累积发生率仍然高达9%以上[47,48]。
多年来的临床研究结果显示:IFNα治疗包括PEGIFNα治疗,可以极大降低慢乙肝患者的肝癌发生风险,5年的肝癌发生风险基本为1%左右,有的甚至仅有0%[25-27,54-57]。近来年多项比较(PEG)IFNα与NA降低肝癌风险作用的研究均发现:PEGIFNα降低慢乙肝患者肝癌风险显著优于NA[25-27]。
参考文献:
[1]侯金林,魏来,王贵强,etal.乙型肝炎临床治愈:共识与争议[J].中华肝脏病杂志,2020,28(08):636-639.
[2]ChineseSocietyofInfectiousDiseasesCMA,ChineseSocietyofHepatologyCMA.GuidelinesforthepreventionandtreatmentofchronichepatitisB(version2019)[J].JClinHepatol,2019,35(12):35(12).
[3]KumadaT,ToyodaH,TadaT,etal.Effectofnucleos(t)ideanaloguetherapyonhepatocarcinogenesisinchronichepatitisBpatients:apropensityscoreanalysis[J].JHepatol,2013,58(3):427-433.
[4]HosakaT,SuzukiF,KobayashiM,etal.Long-termentecavirtreatmentreduceshepatocellularcarcinomaincidenceinpatientswithhepatitisBvirusinfection[J].Hepatology,2013,58(1):98-107.
[5]LiuK,ChoiJ,LeA,etal.Tenofovirdisoproxilfumaratereduceshepatocellularcarcinoma,decompensationanddeathinchronichepatitisBpatientswithcirrhosis[J].AlimentPharmacolTher,2019,50(9):1037-1048.
[6]IkedaK,SaitohS,SuzukiY,etal.InterferondecreaseshepatocellularcarcinogenesisinpatientswithcirrhosiscausedbythehepatitisBvirus:apilotstudy[J].Cancer,1998,82(5):827-835.
[7]ChineseSocietyofHepatologyCMA,ChineseSocietyofInfectiousDiseasesCMA,HouJL,etal.[TheguidelineofpreventionandtreatmentforchronichepatitisB:a2015update][J].ZhonghuaGanZangBingZaZhi,2015,23(12):888-905.
[8]HanM,JiangJ,HouJ,etal.SustainedimmunecontrolinHBeAg-positivepatientswhoswitchedfromentecavirtherapytopegylatedinterferon-alpha2a:1yearfollow-upoftheOSSTstudy[J].AntivirTher,2016,21(4):337-344.
[9]WuY,LiuY,LuJ,etal.DurabilityofInterferon-inducedHepatitisBSurfaceAntigenSeroclearance[J].ClinGastroenterolHepatol,2020,18(2):514-516e512.
[10]LiMH,YiW,ZhangL,etal.PredictorsofsustainedfunctionalcureinhepatitisBenvelopeantigen-negativepatientsachievinghepatitisBsurfaceantigenseroclearancewithinterferon-alpha-basedtherapy[J].JViralHepat,2019,26Suppl1:32-41.
[11]LiuY,LiH,YanX,etal.Long-termefficacyandsafetyofpeginterferoninthetreatmentofchildrenwithHBeAg-positivechronichepatitisB[J].JViralHepat,2019,26Suppl1:69-76.
[12]ChuangWL,JiaJ,ChanHLY,etal.Responsesaredurableforupto5yearsaftercompletionofpeginterferonalfa-2atreatmentinhepatitisBeantigen-positivepatients[J].AlimentPharmacolTher,2018,47(9):1306-1316.
[13]vanZonneveldM,HonkoopP,HansenBE,etal.Long-termfollow-upofalpha-interferontreatmentofpatientswithchronichepatitisB[J].Hepatology,2004,39(3):804-810.
[14]SunJ,DingH,ChenG,etal.SustainedserologicalandcompleteresponsesinHBeAg-positivepatientstreatedwithPeginterferonalfa-2b:a6-yearlong-termfollow-upofamulticenter,randomized,controlledtrialinChina[J].BMCGastroenterol,2019,19(1):65.
[15]WangGQ,HouFQ,ZhangM.LONG-TERMFOLLOW-UPOFPEGINTERFERONΑLFATREATMENTOFHBEAG-POSITIVECHRONICHEPATITISBPATIENTSINPHASEIIISTUDYOFPEGINTERFERONΑLFA-2b(40kD,YSHAPE)(PEGBERON):A4-YEARFOLLOW-UPREPORT[J].Hepatology,2020,AASLD2020Abstract(Poster801).
[16]LeeSK,KwonJH,LeeSW,etal.SustainedOffTherapyResponseafterPeglyatedInterferonFavorsFunctionalCureandNoDiseaseProgressioninChronicHepatitisB[J].LiverInt,2020.
[17]LiuF,WangXW,ChenL,etal.Systematicreviewwithmeta-analysis:developmentofhepatocellularcarcinomainchronichepatitisBpatientswithhepatitisBsurfaceantigenseroclearance[J].AlimentPharmacolTher,2016,43(12):1253-1261.
[18]JiangJF,SunJ,ShiJ,etal.Letter:hepatocellularcarcinomariskafterhepatitisBsurfaceantigenseroclearance[J].AlimentPharmacolTher,2017,45(9):1286-1288.
[19]YipTC,ChanHL,WongVW,etal.ImpactofageandgenderonriskofhepatocellularcarcinomaafterhepatitisBsurfaceantigenseroclearance[J].JHepatol,2017,67(5):902-908.
[20]TatedaK,SuzukiF,KobayashiM.PredictiveFactorsAssociatedwithHepatocellularCarcinomaIncidenceandMortalityafterHepatitisBSurfaceAntigenSeroclearanceinPatientswithChronicHepatitisB[J].Journalofhepatology,2018,AASLD2018abstract(oral213).
[21]HannahSC,RyanA,OliverL.AssociationbetweenHBsAglossandriskofhepatocellularcarcinomainchronichepatitisB:asystematicreviewandMeta-analysis.[J].Journalofhepatology,2019,AASLD2019.Abstracts(poster671).
[22]LokAS,PerrilloR,LalamaCM,etal.medLowIncidenceofAdverseOutcomesinAdultswithChronicHepatitisBVirusInfectionintheEraofAntiviralTherapy[J].Hepatology,2020.
[23]AndersonRT,ChoiHSJ,LenzO,etal.AssociationBetweenSeroclearanceofHepatitisBSurfaceAntigenandLong-TermClinicalOutcomesofPatientsWithChronicHepatitisBVirusInfection:SystematicReviewandMeta-Analysis[J].ClinGastroenterolHepatol,2020.
[24]DongY,LiM,ZhuS,etal.Denovocombinationantiviraltherapyineantigen-negativechronichepatitisBvirus-infectedpaediatricpatientswithadvancedfibrosis[J].JViralHepat,2020.
[25]LiSY,LiH,XiongYL,etal.PeginterferonispreferabletoentecavirforpreventionofunfavourableeventsinpatientswithHBeAg-positivechronichepatitisB:Afive-yearobservationalcohortstudy[J].JViralHepat,2017,24Suppl1:12-20.
[26]RenP,CaoZ,MoR,etal.Interferon-basedtreatmentissuperiortonucleos(t)ideanaloginreducingHBV-relatedhepatocellularcarcinomaforchronichepatitisBpatientsathighrisk[J].ExpertOpinBiolTher,2018,18(10):1085-1094.
[27]LiangKH,HsuCW,ChangML,etal.PeginterferonIsSuperiortoNucleos(t)ideAnaloguesforPreventionofHepatocellularCarcinomainChronicHepatitisB[J].JInfectDis,2016,213(6):966-974.
[28]ChiH,WongD,PengJ,etal.DurabilityofResponseAfterHepatitisBSurfaceAntigenSeroclearanceDuringNucleos(t)ideAnalogueTreatmentinaMultiethnicCohortofChronicHepatitisBPatients:ResultsAfterTreatmentCessation[J].ClinInfectDis,2017,65(4):680-683.
[29]LokAS,ZoulimF,DusheikoG,etal.DurabilityofHepatitisBSurfaceAntigenLossWithNucleotideAnalogueandPeginterferonTherapyinPatientsWithChronicHepatitisB[J].HepatolCommun,2020,4(1):8-20.
[30]AlawadAS,AuhS,SuarezD,etal.DurabilityofSpontaneousandTreatment-RelatedLossofHepatitisBsAntigen[J].ClinGastroenterolHepatol,2020,18(3):700-709e703.
[31]YipTC,WongGL,WongVW,etal.DurabilityofhepatitisBsurfaceantigenseroclearanceinuntreatedandnucleos(t)ideanalogue-treatedpatients[J].JHepatol,2017.
[32]JengWJ,ChenYC,ChienRN,etal.IncidenceandpredictorsofhepatitisBsurfaceantigenseroclearanceaftercessationofnucleos(t)ideanaloguetherapyinhepatitisBeantigen-negativechronichepatitisB[J].Hepatology,2018,68(2):425-434.
[33]ButiM,WongDK,GaneE,etal.SafetyandefficacyofstoppingtenofovirdisoproxilfumarateinpatientswithchronichepatitisBfollowingatleast8yearsoftherapy:aprespecifiedfollow-upanalysisoftworandomisedtrials[J].LancetGastroenterolHepatol,2019,4(4):296-304.
[34]CaoJ,ChiH,YuT,etal.Off-TreatmentHepatitisBVirus(HBV)DNALevelsandthePredictionofRelapseAfterDiscontinuationofNucleos(t)ideAnalogueTherapyinPatientsWithChronicHepatitisB:AProspectiveStopStudy[J].JInfectDis,2017,215(4):581-589.
[35]SuTH,YangHC,TsengTC,etal.DistinctRelapseRatesandRiskPredictorsAfterDiscontinuingTenofovirandEntecavirTherapy[J].JInfectDis,2018,217(8):1193-1201.
[36]HadziyannisSJ,SevastianosV,RaptiI,etal.SustainedresponsesandlossofHBsAginHBeAg-negativepatientswithchronichepatitisBwhostoplong-termtreatmentwithadefovir[J].Gastroenterology,2012,143(3):629-636e621.
[37]BergT,SimonKG,MaussS,etal.Long-termresponseafterstoppingtenofovirdisoproxilfumarateinnon-cirrhoticHBeAg-negativepatients-FINITEstudy[J].JHepatol,2017,67(5):918-924.
[38]ChenCH,HungCH,WangJH,etal.Long-termincidenceandpredictorsofhepatitisBsurfaceantigenlossafterdiscontinuingnucleosideanaloguesinnoncirrhoticchronichepatitisBpatients[J].ClinMicrobiolInfect,2018,24(9):997-1003.
[39]LiuF,LiuZR,LiT,etal.Varying10-yearoff-treatmentresponsestonucleos(t)ideanaloguesinpatientswithchronichepatitisBaccordingtotheirpretreatmenthepatitisBeantigenstatus[J].JDigDis,2018,19(9):561-571.
[40]ChiH,HansenBE,YimC,etal.Reducedriskofrelapseafterlong-termnucleos(t)ideanalogueconsolidationtherapyforchronichepatitisB[J].AlimentPharmacolTher,2015,41(9):867-876.
[41]HungCH,WangJH,LuSN,etal.HepatitisBsurfaceantigenlossandclinicaloutcomesbetweenHBeAg-negativecirrhosispatientswhodiscontinuedorcontinuednucleosideanaloguetherapy[J].JViralHepat,2017,24(7):599-607.
[42]YaoCC,HungCH,HuTH,etal.IncidenceandpredictorsofHBVrelapseaftercessationofnucleosideanaloguesinHBeAg-negativepatientswithHBsAg</=200IU/mL[J].SciRep,2017,7(1):1839.
[43]MaTL,HuTH,HungCH,etal.IncidenceandpredictorsofretreatmentinchronichepatitisBpatientsafterdiscontinuationofentecavirortenofovirtreatment[J].PLoSOne,2019,14(10):e0222221.
[44]LiemKS,FungS,WongDK,etal.Limitedsustainedresponseafterstoppingnucleos(t)ideanaloguesinpatientswithchronichepatitisB:resultsfromarandomisedcontrolledtrial(TorontoSTOPstudy)[J].Gut,2019,68(12):2206-2213.
[45]WongGL,YuenBW,HuiVWK.CommonhepatitisflarebutrareHBsAgseroclearanceafterstoppingNAtreatmentinpatientswithHBeAg-negativeCHBpatients[J].Journalofhepatology,2019,AASLD2019.Abstracts(poster474).
[46]KumarK,SarinSK,SharmaMK.TostudytheefficacyifPEG-IFNαHBeAgnegativechronichepatitisBpatientsafterstoppingnuvleotideanaloguetherapy:clinicaltrial.GOVidentifiedNO:NCT03123653[J].Journalofhepatology,2019,AASLD2019.Abstracts(poster480).
[47]KimSU,SeoYS,LeeHA,etal.HepatocellularCarcinomaRiskSteadilyPersistsoverTimeDespiteLong-TermAntiviralTherapyforHepatitisB:AMulticenterStudy[J].CancerEpidemiolBiomarkersPrev,2020,29(4):832-837.
[48]SouFM,HuTH,HungCH,etal.Incidenceandpredictorsofhepatocellularcarcinomabeyondyear5ofentecavirtherapyinchronichepatitisBpatients[J].HepatolInt,2020,14(4):513-520.
[49]WangJP,KaoFY,WuCY,etal.Nucleos(t)ideanaloguesassociatedwithareducedriskofhepatocellularcarcinomainhepatitisBpatients:apopulation-basedcohortstudy[J].Cancer,2015,121(9):1446-1455.
[50]WuCY,LinJT,HoHJ,etal.Associationofnucleos(t)ideanaloguetherapywithreducedriskofhepatocellularcarcinomainpatientswithchronichepatitisB:anationwidecohortstudy[J].Gastroenterology,2014,147(1):143-151e145.
[51]ChoJY,PaikYH,SohnW,etal.PatientswithchronichepatitisBtreatedwithoralantiviraltherapyretainahigherriskforHCCcomparedwithpatientswithinactivestagedisease[J].Gut,2014,63(12):1943-1950.
[52]KimSU,SeoYS,LeeHA,etal.Amulticenterstudyofentecavirvs.tenofovironprognosisoftreatment-naivechronichepatitisBinSouthKorea[J].JHepatol,2019,71(3):456-464.
[53]HsuYC,WongGL,ChenCH,etal.TenofovirVersusEntecavirforHepatocellularCarcinomaPreventioninanInternationalConsortiumofChronicHepatitisB[J].AmJGastroenterol,2020,115(2):271-280.
[54]LinSM,SheenIS,ChienRN,etal.Long-termbeneficialeffectofinterferontherapyinpatientswithchronichepatitisBvirusinfection[J].Hepatology,1999,29(3):971-975.
[55]LeeD,ChungYH,LeeSH,etal.Effectofresponsetointerferon-alphatherapyontheoccurrenceofhepatocellularcarcinomainpatientswithchronichepatitisB[J].DigDis,2012,30(6):568-573.
[56]PapatheodoridisGV,ManesisE,HadziyannisSJ.Thelong-termoutcomeofinterferon-alphatreatedanduntreatedpatientswithHBeAg-negativechronichepatitisB[J].JHepatol,2001,34(2):306-313.
[57]LinSM,YuML,LeeCM,etal.InterferontherapyinHBeAgpositivechronichepatitisreducesprogressiontocirrhosisandhepatocellularcarcinoma[J].JHepatol,2007,46(1):45-52.
声明: 不要轻信任何医疗广告或新闻报道式广告! 请勿轻信素未谋面的网络医生! 本站所刊载的信息仅供参考,不能代表医生的诊断和治疗,请勿直接对照治疗而延误病情! 因个人主职工作时间不宽裕,网站未来仅保持不定期更新。









全部评论:暂无评论